During in vitro culture of human embryos, there are several important fundamentals that need to be measured and controlled. Read this first in a series of articles addressing the importance of quality control and measurements of the culture environment, for the optimal development of embryos and success in the IVF laboratory.
Steve Levett PhD, our Director of Clinical Application, addresses the importance of culturing at optimal pH and maintaining a stable environment.
Most embryologists recognize that they should measure the pH of the culture media in their culture system but most struggle to find an accurate, dependable and reproducible way of testing it. This is especially true when you introduce oil into the equation.
Consequently, most embryologists do not measure the pH of their media but rely, instead, on measuring CO2 concentration in each incubator. Stable CO2 concentration should lead to a stable pH environment but the only way to know for sure is to measure pH.
What is pH?
pH is a measure of acidity or alkalinity expressed by the hydrogen ion concentration. Acids are defined as substances that increase H+ concentration and bases are things that decrease this concentration. The pH scale ranges from 1 to 14. Substances with a pH of less than 7 are considered acidic, substances with a pH greater than 7 are basic. pH of 7 is considered neutral.
Most embryo media utilize a bicarbonate/CO2 buffer system to maintain a physiological pH of 7.2-7.4 in the culture medium. The balance between the CO2 concentration in the incubator and the bicarbonate concentration in the culture media determines the pH that the embryo is exposed to.
Why is pH important?
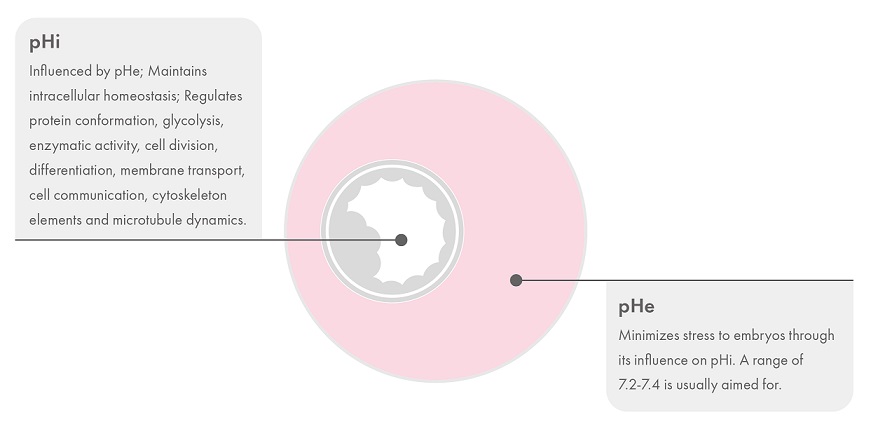
The intracellular pH (pHi) of oocytes and embryos plays an essential role in maintaining homeostasis and regulating several critical metabolic and transport processes, including protein conformation and glycolysis.1 In human embryos, the pHi initially follows the external pH of the culture environment (pHe) and any fluctuations in the pHe will, therefore, affect the embryo’s development.2
Embryos do possess mechanisms to regulate pHi, however, this is an energy-requiring process2 and has an impact on the cellular resources expended.3 A study on hamster embryos has shown that slightly raising or lowering the pH for a few hours results in the localization of mitochondria and actin cytoskeletal elements.4 Later stages of embryos regulate their pHi more efficiently than cleavage stage embryos, which is likely due to the latter having tighter cell junctions. Warmed, vitrified embryos lack the ability to modulate their pHi for several hours post-warming.5
Denuded mature oocytes also lack the ability to regulate pHi and are consequently dependent upon the pH of the culture environment. It is therefore important to avoid pH fluctuations during oocyte handling and manipulation. As the oocyte’s cytoskeleton is involved in the positioning of the meiotic spindle6 and, as described above, pH is known to affect similar actin cytoskeletal elements in the embryo,4 procedures like ICSI, where the protective cumulus cells are removed, could create embryos with chromosomal abnormalities.3
What is the optimal pH?
The optimal pH has not been accurately determined for human embryo culture but in general, a physiological range of 7.2-7.4 is the aim. Embryo pHi is approximately 7.1 and using a pHe too far away from the pHi value may stress the embryo as more resources are required to maintain homeostasis. Consequently, the pHe should be slightly higher than the pHi to help offset the acidification that occurs as a result of intracellular metabolic processes.
How do you get the optimal pH in your lab?
Setting the correct, optimal, pH of culture media depends on the relationship between the concentration of bicarbonate in the culture media itself and the CO2 concentration in the laboratory incubator. This, in turn, is affected by atmospheric pressure.
There is an inverse relationship between CO2 concentration and pH. Adjusting the CO2 concentration in the incubator changes the pH of the culture media: so to decrease pH you need to increase CO2 concentration and vice versa. The relationship depends on the concentration of sodium bicarbonate, NaHCO3 in the media, which will dissociate according to the equation below.2
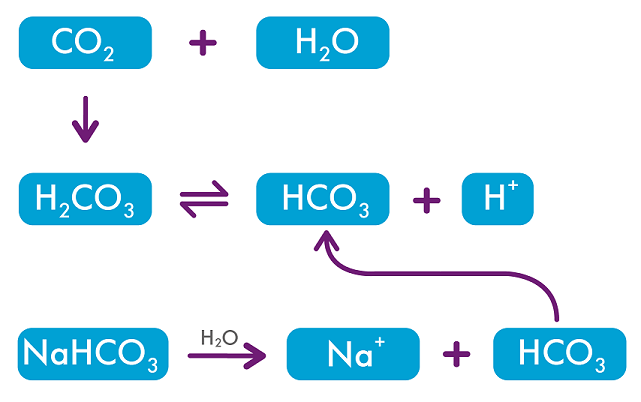
To determine what CO2 concentration to use, the pH of the culture media must be measured and adjustments made based on these findings.
Accurate measurement of CO2 concentration in the incubator is critical to maintaining a stable pH environment. The CO2 concentration must be measured by an external, calibrated CO2 gas analyzer. Relying on the incubator’s display is not sufficient.
How can we help your lab?
I spend a lot of time traveling around the world visiting IVF labs to carry out pH testing for them. We have developed a reliable, verified methodology for measuring pH from equilibrated dishes where the media sample is overlaid with oil. This gives the closest estimation of the pH in the media the embryos are cultured in.
This is a service that the Medical Affairs team at CooperSurgical can offer to your lab. If you are interested or would like more information, please contact me by email on [email protected].
Further Reading: Expert Article
Stephen Troup PhD, UK Consultant Reproductive Scientist, has written an article IVF culture media – is it time to go back to basics? in the June/July edition of our scientific newsletter – ART Scientific.
To read the article – Subscribe Here
ART Scientific is a quarterly publication that explores current topics of interest in our industry. It is written by a community of experts in the field and has a science focus at its heart.

Steven Levett PhD
Dr Levett took up his first embryology role in the early 1990s at St James’s University Hospital in Leeds, UK. He became Lab Manager soon after and went on to manage other embryology labs in the UK and Canada, in the public and private sector.
From 2006-2014 Steve worked for Wallace, then part of Smiths Medical, becoming Global Business Director in 2010. He then joined Ferring in 2014 as UK National Business Manager for Obstetrics and Fertility and 18 months later joined Lifeglobal as Vice President, International Sales.
Steve has a keen interest in laboratory optimization, quality control procedures, optimization of results and supporting embryologists. His research interest has been in fertility preservation since working with Professor Roger Gosden in Leeds.

 My Clinic is in the United States
My Clinic is in the United States My Clinic is in Canada
My Clinic is in Canada
