
Earlier this year, specialist groups from ESHRE and Alpha met to discuss indicators for use in monitoring ART lab performance.
The resulting Vienna consensus report recommended twelve key performance indicators (KPIs) that can be used to help laboratories measure their overall clinic performance.
The ability to track various areas of lab performance, and possible contributing factors, is therefore important if clinics are to continue exceeding competency and benchmark levels.
ART Quality management system
RI Witness is an ART management system that helps prevent potential errors and manages activities within IVF laboratories. Using RFID (radio frequency identification) technology, it can be used as a versatile quality control tool to oversee large or small teams, high volumes of work, data collection, auditing, and accountability.
The system has an analytics function that enables you to dig deeper into numbers and statistics, providing a detailed picture of how you can improve your IVF lab quality control. The incorporation of a workflow management module allows individual performance records within a clinic to be analyzed and compared against each other.
The software enables analysis of performance indicators (PIs) related to timings and duration of procedures such as denudation, insemination, fertilization check, media changes and morphological analysis of embryos.
As it is a bespoke system, tailored to suit each clinic’s standard operating procedures (SOPs), the following examples illustrate a selection of ways the analytics function can be used to your advantage.
Track procedure times
Identifying how long it takes for staff members to undertake particular procedures may help uncover a contributing factor towards a drop in fertilization rates, for example:
If expected procedure times are exceeded, RI Witness can flag which operators may require extra training.
A follow-up action could be the reallocation of a more even workload among the team, in terms of procedure numbers and type, ensuring all staff members are given the opportunity to upkeep their skill set.

Example shows procedure times may exceed those deemed appropriate for undertaking ICSI Prep.
The RI Witness analytics function also allows you to track other procedural times including how long it takes staff members to denude, and the time it takes doctors or clinicians to carry out egg collection.
In either situation, if further training has been identified and undertaken, RI Witness can track your staffs’ ongoing improvements and performance.
In the first edition of our new newsletter, ART Scientific, Steven Fleming PhD reviews KPIs and the benchmarking of staff and clinic performance. Click here to subscribe and download your copy today
Identify equipment issues
If your lab is experiencing a dip in blastocyst rates, a contributing factor could be a problematic work area. Reviewing how long procedures take by location, may help identify trends.
In this example, total procedure times are longer on the Embryology 1 workstation in comparison with the Embryology 2 workstation. This may indicate the need to undertake quality checks regarding temperature, VOCs and air quality.
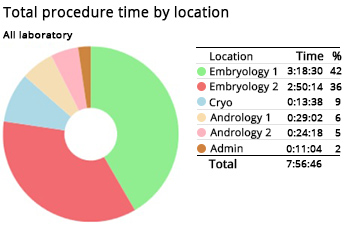
Report shows Embryology 1 workstation may require a quality check due to slower procedure times.
Seeing a drop in blastocyst rates in your clinic database could give rise to further investigation through RI Witness Analytics. For example, you can use location reports to check which workstations were used for particular patients.
Additional factors that can often go unnoticed, such as an underperforming micromanipulator or laser due to damage, can also be identified using the data contained in this type of report. The necessary steps can then be taken to have them repaired by organizing a maintenance or service visit.
With ever-increasing demands on the lab, optimal use of all equipment and stations is critical for efficient workflow. Using the data from a location report, you can discover if a particular workstation is not being used, perhaps due to embryologists’ preferences.
It could be the case that the equipment within it is old and not popular anymore. Alternatively, it may be a new piece of kit that staff members are uncomfortable using as it is unfamiliar or they have not had the appropriate training.
Target workflow disruption
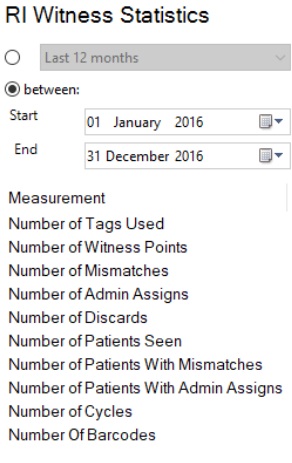
There are also a number of measurements you can track in the Statistic report that can trigger the need for further investigations.
One of these includes the number of Admin Assigns that occur, ie where a manual over-ride of the system has taken place, indicating a break in normal workflow (SOP).
Examples could include patients forgetting RFID cards, an accidental discard or dropped dish, a button pressed mistakenly. Every time a manual over-ride occurs, a reason must be entered into the system – and it is here a Lab Manager may spot trends.
If there are a number of incidents, or the system is being used incorrectly, within a given time period this could indicate staff members are experiencing stress, fatigue, or are struggling with a heavy workload. Alternatively, it may indicate a need for further training.
You can also use the Statistics report to track the number of RFID tags and cryo barcodes used on your plasticware, to help with ordering and setting budgets.
Monitor sample mismatches
RI Witness can track the human error rate in a lab. Looking at the number of mismatches that occur, and the given reason for each mismatch can help identify causes.
Labs, such as IVI UK, are using the system to track human error as a KPI in their lab, alongside using it for other reporting and auditing purposes.
Dr Stephen Troup’s team at IVI UK use RI Witness to track sample mismatches.
Enforce clinic SOPs
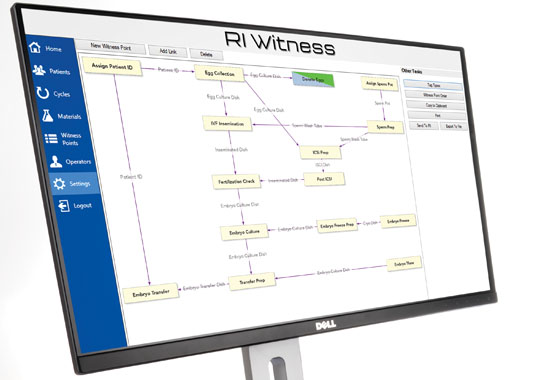
The Witness Point Diagram is a form of process mapping that enables the identification of all meaningful parameters within a clinic.
When setting up RI Witness within a laboratory, this diagram is created with you to exactly mirror individual clinic SOPs. Once set, it helps enforce these SOPs by guiding staff members, requiring them to follow the correct protocols before moving to the next step in a procedure.
ART Scientific Newsletter
Our Director of Embryology, Steven Fleming PhD, has written a review of benchmarking ART laboratories, staff and clinic performance in the November edition of our scientific newsletter – ART Scientific.
To read the article – Subscribe Here
ART Scientific is a quarterly publication that explores current topics of interest in our industry. It is written by a community of experts in the field, and has a science focus at its heart.

 My Clinic is in the United States
My Clinic is in the United States My Clinic is in Canada
My Clinic is in Canada